Water loss and the atmosphere
Plants lose moisture by transpiration from their leaves into
the surrounding atmosphere. The less moisture they lose, the more
they like it. We tend to think that the higher the relative
humidity, the moister the air and the better it is for plants up to
a certain point. The point is that relative humidity does NOT relate
directly to the rate at which transpiration of water from the plant
occurs. Relative humidity is not proportional to the rate of
moisture loss from the plant. How come?
The moisture holding capacity of air is measured in units of
pressure, and there are two important measurements concerned with
figuring out how much moisture a given block of air can potentially
absorb. First is the
saturation vapor pressure (SVP): think of this as the maximum amount
of water vapor a given block of air can hold. Second measurement is the difference between the amount of
water vapor actually in a given block of air and its SVP (i.e., the
maximum amount of water it could absorb). This difference is called the vapor pressure
deficit, or VPD. Think
of VPD as the water sucking power of the air, because it is actually
the VPD that interests your plants, not the relative humidity.
At face value, VPD (sucking power) seems to be the same as
relative humidity - because relative humidity is the ratio of the
actual vapor pressure in the air to the SVP. Its not the same, because the SVP of a given block of air
increases exponentially as the air temperature rises - the higher
the temperature, the greater the amount of water vapor that air can
hold. Rather than giving a physical explanation of why humidity and
VPD are different measurements, because I'll get out of my depth in
about two seconds, just look at how the VPD (sucking power) changes
at various temperatures if the relative humidity stays the same at
75%:
temperature
|
humidity
|
SVP
|
vpd
|
10
|
75
|
1.23
|
0.31
|
11
|
75
|
1.32
|
0.34
|
12
|
75
|
1.40
|
0.35
|
13
|
75
|
1.50
|
0.37
|
14
|
75
|
1.60
|
0.40
|
15
|
75
|
1.70
|
0.43
|
16
|
75
|
1.82
|
0.45
|
17
|
75
|
1.94
|
0.48
|
18
|
75
|
2.06
|
0.52
|
19
|
75
|
2.20
|
0.55
|
20
|
75
|
2.34
|
0.58
|
21
|
75
|
2.49
|
0.62
|
22
|
75
|
2.64
|
0.66
|
23
|
75
|
2.81
|
0.70
|
24
|
75
|
2.98
|
0.75
|
25
|
75
|
3.17
|
0.79
|
26
|
75
|
3.36
|
0.84
|
27
|
75
|
3.57
|
0.89
|
28
|
75
|
3.78
|
0.94
|
29
|
75
|
4.01
|
1.00
|
30
|
75
|
4.24
|
1.06
|
31
|
75
|
4.49
|
1.12
|
32
|
75
|
4.76
|
1.19
|
33
|
75
|
5.03
|
1.26
|
34
|
75
|
5.32
|
1.33
|
35
|
75
|
5.63
|
1.41
|
36
|
75
|
5.95
|
1.49
|
37
|
75
|
6.28
|
1.57
|
38
|
75
|
6.63
|
1.66
|
39
|
75
|
7.00
|
1.75
|
40
|
75
|
7.39
|
1.85
|
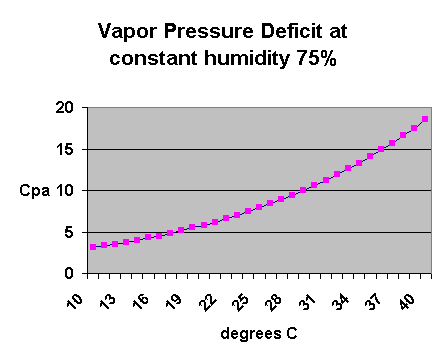
Looking at the temperature and vpd on a graph,
you can see how the vpd is increasing exponentially as the
temperature rises, while the relative humidity remains constant:
In the majority of plants
atmospheric carbon dioxide is captured through tiny pores in the
epidermis (called stomata) that open during the daylight hours
(called C3 respiration). Oxygen produced as a byproduct is expelled.
If the outside air is dryer than the epidermal cells, which it
usually is during sunlight hours, water vapor will diffuse out into
the atmosphere through the open stomata. This moisture loss is a
disadvantage for the plant, which usually has to take up
compensatory water through its root system.
That's the "standard model"
of plant behavior. Its not, in fact, how XTs normally operate. Moisture loss is a huge problem for XTs, because they do not
have access to root-derived water. They have evolved with a different method of capturing
atmospheric carbon dioxide. This enables them to minimize water loss
by using a process called CAM respiration. Unlike the "standard model plant," during the day, the XTs'
leaf pores, the stomata, close up; thereby avoiding moisture loss to
a considerable degree. Through the night hours, when the temperature
drops and the VPD decreases (hereby reducing the potential for water
vapor loss from the plant), the air has much less sucking power and
the CAM plant opens its stomata and captures carbon dioxide. As the
carbon dioxide is captured it is converted to organic compounds -
mainly malic acid - which are stored until daylight. Around dawn,
the stomata close up again. During the day, stored malic acid is
converted back to carbon dioxide within the plant tissues and the
Calvin Benson cycle proceeds to fix carbon and the plant produces
its energy stores. [3]
Remember the mechanics of
vapor pressure deficit, VPD. It is a measure of the capacity, or potential, of air to take
up more moisture. The higher the VPD the greater the potential for
sucking moisture out of the plant. As we saw, when the temperature rises, air round your plant
sucks moisture from open stomata faster and faster unless the
relative humidity increases at a rate corresponding to the
increasing temperature. On the other hand, as the temperature drops the moisture-holding
capacity decreases exponentially. So a cool night equals a lowering of the VPD, much lower
moisture loss for the XT while its stomata are open to receive
carbon dioxide (remember that deserts generally have very cool
nights. The only one I have spent the night in was the Makgadikigadi
Pans in Botswana, and it was COLD.)
One of the places people have
difficulty growing XTs is in the home. I think this is particularly
true if you have an air-conditioned house. Look at what happens in
our office. The room is
about 150 m3 and the air-conditioner is a Fujitsu reverse cycle
unit.
 |
Without the air-conditioner,
the VPD is running between 5 and 6 at night, climbing to about 10
during the day. Once we turn on the air-conditioner the VPD jumps up
to between 20 and 25 while the thing is on. At that sort of range
plants are going to start sucking up a lot of water to keep up, and
I doubt that XTs will cope for long. This is a good indicator of why
watering requirements for XTs are going to be much different in the
two types of situation. I don't grow XTs indoors, but one of our
Wellington Tillandsia Study Group members, Beryl McKellar, does[4]. We can assume that her conditions were very similar to my
house without air-conditioning. She grows her plants indoors, as I
said, and mists them every morning in summer, every 3 to 4 days in
winter. But in the drier,
high-deficit air of the air-conditioned room, you would surely have
to soak the plants regularly to make up for the water loss they
suffer.
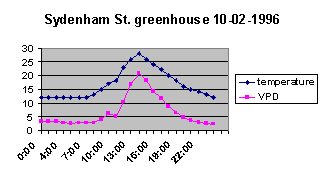
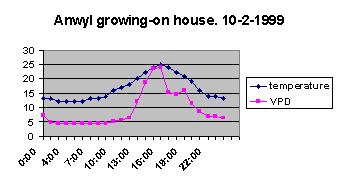
The night-time VPD is still down at the 4 to 5 level, and I
would say that this is probably around the maximum you would want to
sustain for mature XTs. Let's use it as a starting figure, anyway. I have no idea how
sensitive XTs are to VPD range at night, but my experience shows
that young seedlings are very sensitive. To recap, I start XT seeds
off, and grow them for up to nine months, in my incubator. This is a
solid timber box; 1200mm long, 700mm high and 700mm deep. Two fluorescents are hung in the ceiling, immediately below
them is a wire-netting screen from which the seeds are sown on
hanging strips of fine plastic netting. In the bottom is a deep tray
with wet sand in it covering a soil-warming cable. The front of the
box was originally a solid wood panel that lifted up to allow
access. This incubator was always very successful, and particularly
I never lost any seedlings once they got started. However, about a
year ago I replaced the solid front panel with a double thickness of
plastic so my mother-in-law could get at the seeds to water while we
were away. And since then, I have had a lot of problems including
the loss of about 60 batches of seedlings last year. Previously, I
only had to water the seedlings once or twice a day; now I have to
water 4 times a day or I start losing them. Given that the light and temperature are pretty much the same
in both cases, look at how the VPD differs:
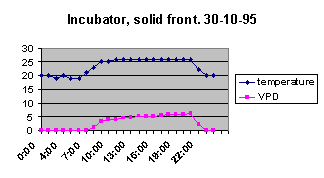
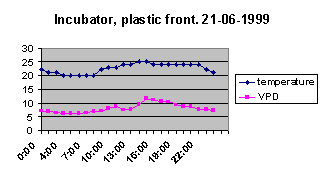
In the solid incubator, the relative humidity
stayed at 100% while the lights were off—in the plastic fronted
version it only gets to 75%. Whilst 75% relative humidity sounds
pretty good, look at the difference in VPD rates! In effect, then,
seedlings thrive when the VPD ranges between 0 and 5 Cps, but are
very difficult and require heavy watering when the VPD stays between
5 and 10 Cps.
Conclusion
It is far too early for me to try laying down
firm guidelines for what sort of VPD ranges we should be looking for
at night. There are many other factors of at least equal importance
to get right, particularly temperature and light. Studying the VPD
does not negate the standard advice that the higher the temperature,
the higher you need the relative humidity. But it does start to show
us how to quantify the actual relative humidity we should be looking
to provide, especially at night. It helps us understand why the cool
nights seem to be required, to get the VPD down and avoid excess
moisture loss while the XTs are taking up carbon dioxide.
Measuring VPD can be a valuable tool to assist us
in deciding how much to water our tillandsias. And it can also guide
us to whether they are in need of more, or less, of the fresh moving
stuff.
2. Look up the SVP of air at that
temperature (Celsius) by looking at the following table:
Temperature
|
SVP
|
1
|
0.657421947513385
|
2
|
0.706315438214953
|
3
|
0.75839908974603
|
4
|
0.813850507765805
|
5
|
0.8728553593282
|
6
|
0.935607645775277
|
7
|
1.00230998096997
|
8
|
1.07317387484204
|
9
|
1.14842002221719
|
10
|
1.22827859689512
|
11
|
1.31298955093833
|
12
|
1.40280291912957
|
13
|
1.49797912855174
|
14
|
1.59878931324
|
15
|
1.70551563385225
|
16
|
1.81845160229991
|
17
|
1.93790241127728
|
18
|
2.06418526862391
|
19
|
2.19762973645066
|
20
|
2.33857807495646
|
21
|
2.48738559085903
|
22
|
2.64442099035948
|
23
|
2.81006673655684
|
24
|
2.9847194112255
|
25
|
3.16879008086477
|
26
|
3.3627046669269
|
27
|
3.56690432012621
|
28
|
3.78184579872918
|
29
|
4.00800185072203
|
30
|
4.24586159974958
|
31
|
4.49593093471598
|
32
|
4.75873290293534
|
33
|
5.03480810671739
|
34
|
5.32471510327082
|
35
|
5.62903080780417
|
36
|
5.94835089970204
|
37
|
6.28329023165168
|
38
|
6.63448324159299
|
39
|
7.00258436736282
|
40
|
7.38826846390217
|
3. Calculate the VPD. Subtract the relative humidity from 100, divide that figure
by 100, and then multiply the result by the SVP. Answer is the VPD
in Kilopascals.
Example: 75% RH at 30 C. From table, SVP = 4.245861599.
Subtract RH from 100, equals 25.
25 divided by 100, equals 0.25
.25 times 4.245861599, equals 1.0615 Kpa.
IMPORTANT: Vapor pressure deficit is usually
measured in kilopascals, as shown in this example. However, for the
purpose of my talk I used centipascals, i.e., the Kpa figure
multiplied by 10. I did this so you could see the VPD readings on
the graphs because I could only put one scale on with my stupid
software….. or I was too stupid to figure out how to put two scales
on the graphs. Whatever. I think it is easier to "visualize" numbers
between 0 and 20, than it is with the much smaller variations if
they were between 0 and 2.
NOTES:
1. Duncan Golicher, "tillandsia
growing on-Relative humidity" brom-l@bdt.org.br (21 Jan 1997)
2. Mark A. Dimmitt, Ed. Bromeliads: A Cultural Manual.
The Bromeliad Society Inc., 1992
3. I am not a plant physiologist, or a
biologist! This is drawn from reading David Benzing, The Biology of The Bromeliads (Eureka, Mad River Press, 1980), and my classes a few decades ago in
BOTY111 and BIOL111.
4. Andrew Flower, "Wellington
Tillandsia Study Group"
Bromeliad Society of New Zealand Bulletin 38 #3, April 1998. Page 49